Abstract
CMV is the most clinically significant viral infection in HCT recipients. Control CMV reactivation after HCT is highly dependent on CMV-specific T cells. Despite dramatic technical advances, the clinical utility of functional assays of virus-specific T cells to predict CMV reactivation following HCT remains to be established.
Using 13-color flow cytometry, we studied CD8+T cell responses to pp65 and IE-1 CMV peptide stimulation in cryopreserved PBMC from three clinically distinct subgroups (n=10 each) of HCT patients: 1) Elite Controllers (EC) : CMV seropositive (R+) recipients who never reactivated CMV based on weekly surveillance testing; 2) Spontaneous Controllers (SC): CMV R+ recipients who spontaneously resolved low-grade viremia (<200 IU/mL) without antiviral therapy; and 3) Non-controllers (NC): CMV R+ recipients who experienced high-grade CMV viremia (>1,000 IU/mL) requiring antiviral therapy.
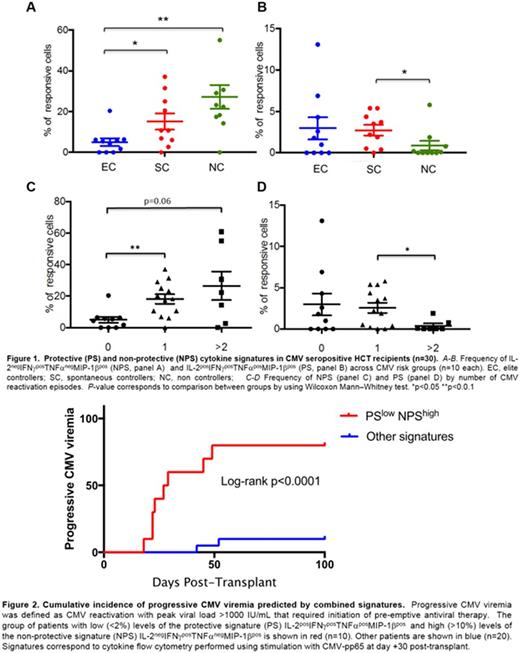
NC had lower numbers of CD8+ T cells that simultaneously produced 3-4 cytokines in response to CMV peptides compared to EC and SC (18, 26 and 34%, respectively) suggesting that progressive CMV viremia is associated with loss of CD8+ T cell polyfunctionality. Among 15 possible cytokine signatures, we identified two CMV-specific CD8+ T cell cytokine signatures, measured at day +30, that were strongly associated with the risk of CMV reactivation (Fig. 1): i) the non-protective signature (NPS) consisting of IL-2negIFNγposTNFαnegMIP-1βpos CD8+ T cells was positively associated with CMV reactivation (4.9% of CMV-specific CD8+ T cells vs. 19.4 P=0.002 for EC vs. SC/NC; 4.9 vs. 10.8 P=0.02 for EC vs. SC; 4.9 vs. 22.8 P=0.005 for EC vs. NC for pp65 stimulated cells; similar trends were observed in IE-1 stimulated cells); ii) the protective signature (PS) consisted of quadruple producers (IL-2posIFNγposTNFαposMIP-1βpos), and was significantly reduced among NC vs. SC following pp65 and IE-1 stimulation (0.05% of CMV-specific CD8+ T cells vs. 2.85 for pp65, P= 0.02; 0 vs. 1.25 for IE-1, P= 0.02); this association was also found in superantigen-stimulated cells. Production of IFNγ alone or in combination did not predict reactivation (P=0.49).
Since NC trended toward more frequent recurrence of CMV viremia compared to SC (60 vs. 10%, respectively; P=0.06), we explored the association between PS and NPS and number of episodes of CMV reactivation (Fig. 1). We observed a significant stepwise increase in the levels of the NPS in pp65-stimulated cells in patients who experienced 0 vs. 1 and ≥2 episodes of CMV reactivation (4.9, 18.3 P=0.002, and 22.4 P=0.06). In addition, patients with ≥2 episodes of CMV had the lowest levels of the PS across groups (0 vs. 2.2% for ≥2 vs. 1 episode P=0.02). Similar trends were observed in IE-1 stimulated cells.
Whereas T-cell depletion, aGVHD, lymphoid malignancy and CMV donor serostatus were not associated with risk of CMV in this small cohort, a NPS >10% was associated with increased risk of CMV reactivation (OR: 21, CI95 2-215; P=0.01) and need for treatment (OR: 14, CI95 1.5-137; P=0.02); and a PS >2% was associated with trend toward reduced risk of need for treatment (OR: 0.1, CI95 0.01-1.05; P=0.06). Multivariable modeling was not performed due to sample size.
Time to event curves showed that high levels of NPS (>10%) predicted risk of CMV reactivation (log-rank P=0.0002). This remained true in analyses restricted to patients with CMV reactivation after day 30 (log-rank P=0.01). High levels of NPS or low levels of PS (<2%) predicted risk of need for treatment (log-rank P=0.003 and P=0.04, respectively). Combination of the PSlow/NPShigh had the highest predictive value for risk of need for treatment (log-rank P <0.0001; fig. 2).
18 graft products were available. As expected CMV-specific responses were not detected in grafts from CMV seronegative donors. Among the 9 CMV seropositive grafts, NPS expression was null across groups suggesting that the NPS is an immune phenotype that is absent in healthy donors; there was a stepwise decrease in the number of quadruple producer CD8+ T cells in CMV seropositive grafts for EC, SC and NC: 3.4 (n=3), 2 (n=4) and 0 (n=2), respectively, suggesting that donor PS might influence recipient reactivation.
In conclusion, we have identified two novel CMV-specific CD8+ T cell cytokine signatures with robust predictive value for risk of CMV reactivation and need for treatment. These biomarkers might be useful in guiding clinical decision making in HCT recipients.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal